Collection: Buffers
UFC Bio’s buffer lineup features precision-grade solutions like 0.9% sterile saline for irrigation wash, alongside PBS, TE, TBE, and HEPES, each crafted sterile and USA-manufactured for reliable lab performance.
Free U.S. shipping over $50
-
TE Buffer, low EDTA, pH 8.0, 500mL
Regular price $40.00 USDRegular priceUnit price / per -
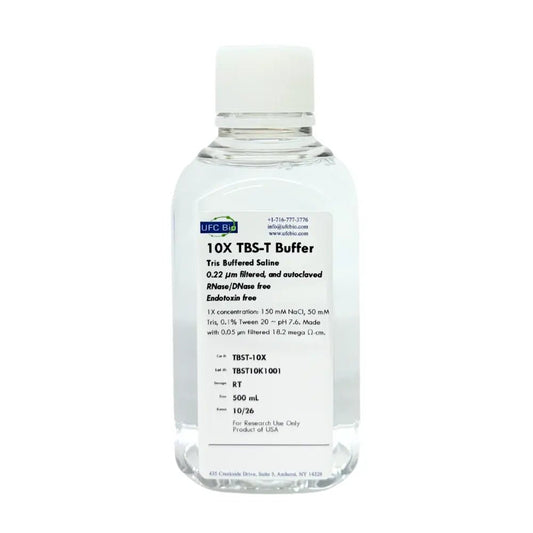
10X TBS-T (Tris-buffered Saline with Tween-20), 500 mL
Regular price $30.00 USDRegular priceUnit price / per -
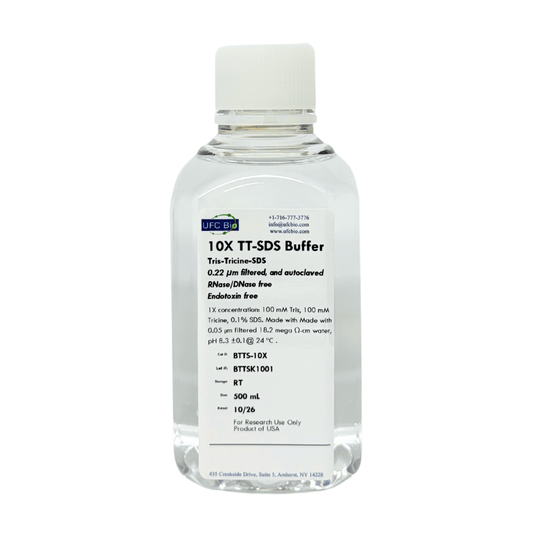
10X Tris-Tricine-SDS
Regular price $49.99 USDRegular priceUnit price / per -
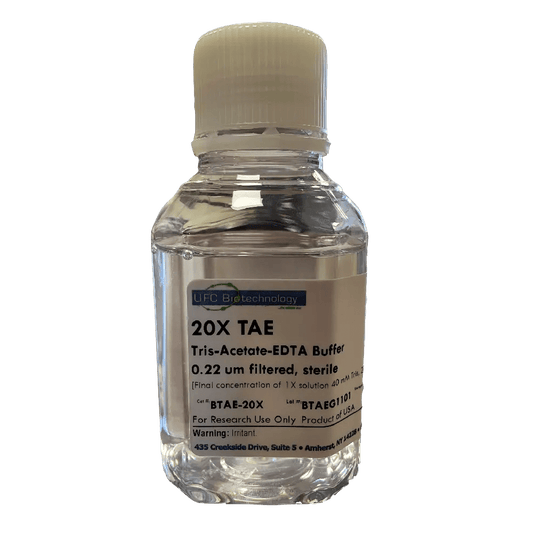
20X TAE (Tris Acetate EDTA) Buffer, 250 mL
Regular price $25.00 USDRegular priceUnit price / per